


Ocular Vascular Clinic
Department of Ophthalmology & Visual Sciences
University of Iowa Carver College of Medicine
Iowa City, Iowa
This is part one in a pair of articles about AION.
The first part of this article is intended primarily for patients and lay people, and the second part (on a separate web page) is intended for ophthalmologists. The article is a summary of material published in peer-reviewed ophthalmic journals. For more detailed information, please refer to the papers in the bibliography and the various articles cited in those papers.
Dr. Hayreh does not give an opinion without personally examining a patient; he feels that to do so is unethical and also potentially dangerous.
Originally published 1995 and revised, expanded and modified in April 2009, reviewed November 2010, February 2013, August 2016.
This article is intended to give basic information about ischemic optic neuropathy, which is sometimes called "stroke of the optic nerve" in lay persons' term. Ischemic optic neuropathy is one of the most common causes of loss of vision in persons past middle age. However, it does occur less commonly, in younger persons. Knowledge about this disease will enable patients and their doctors to work together to understand the disease. Early diagnosis, recognition of various risk factors, and carrying out the treatment and follow-up visits as recommended, are the most important aspects of care for persons with ischemic optic neuropathy. The following description of ischemic optic neuropathy is based on our basic, experimental and clinical studies published in the peer reviewed journals. [1-77]
The eye is often compared to a camera. Light comes in through the cornea, pupil, and lens at the front of the eye just as the lens of the camera lets in light to the film. This light is then focused on the inside wall of the eye called the retina (as on the film in a camera). This picture is then sent to the brain along the optic nerve, which connects the eye to the brain. We see properly when all of these parts of the eye and brain are working together
The optic nerve is unique because it is the only nerve in the body that a physician is able to see directly (without surgery) as it begins at the back of eye. The optic nerve is made up of a million tiny, delicate nerve fibers that are like wires. Many blood vessels nourish the optic nerve with blood rich in oxygen and nutrition. Vision actually takes place in the brain when the messages from the eye travel to the brain along the optic nerve; however, the nerve has to be healthy to transmit these messages. The whole situation may be compared to a video camera (i.e. the eye) taking the picture and transmitting along the cable (the optic nerve) to the television (the brain); if the cable is damaged for any reason, although the video camera and the television may be perfectly normal, a distorted picture or no picture is seen on the television.
While explaining this disease, a doctor may say that there has been a "stroke at the back of the eye", "blocked blood vessel", or "broken blood vessel at the back of the eye". A "stroke" is an interruption of the blood supply to the brain or a nerve in the body. When a person has a major "stroke" in the brain, the blood supply to a part of the brain that controls speech or movement of the arms and legs is often affected. With a "stroke at the back of the eye" a loss of vision develops because the retina or the optic nerve may have its circulation interrupted. How much vision is lost depends on how much the retina or the optic nerve is affected. In this website we are discussing only the stroke of the optic nerve and NOT that of the retina.
Ischemia of the optic nerve results in ischemic optic neuropathy [17, 24, 36, 49, 57, 74]. Ischemia is a decrease in the blood supply to a tissue in the body which in turn decreases oxygen and nutrition. Without sufficient oxygen and nutrition, nerves cannot function properly and will eventually die. As an example, placing a tight rubber band around the wrist causes the fingers to begin to tingle and then lose the sensation of touch because the rubber band cuts off the blood supply, starving the nerves of oxygen and nutrients. If the blood supply to the optic nerve is interrupted, it produces ischemia of the optic nerve (technical name, "ischemic optic neuropathy"), and a loss of vision develops. The blood supply of the anterior (Fig. 1-A) of the optic nerve (also called the optic nerve head) is mainly by the posterior ciliary arteries, while the blood supply to the posterior part (back part of Fig. 1-B) is from several quite different sources but not the posterior ciliary arteries [4-6, 8, 9, 13, 18, 25, 30, 35, 36, 49, 52, 74].
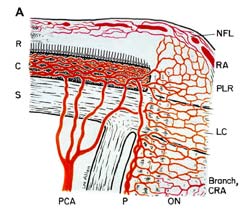
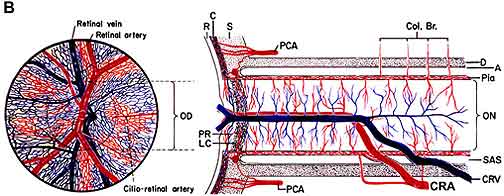
Ischemic optic neuropathy is of two types [17, 36, 74], depending upon the part of the optic nerve involved:
Anterior ischemic optic neuropathy (commonly abbreviated to "AION"): AION is due to acute ischemia of the front (anterior) part of the optic nerve (also called optic nerve head), which is supplied mainly by the posterior ciliary arteries.
Posterior ischemic optic neuropathy (commonly abbreviated to "PION"): PION is a much less common type [24, 60]. It is due to acute ischemia of the back (posterior) part of the optic nerve, located some distance behind the eyeball; this part of the optic nerve is NOT supplied by the posterior ciliary arteries. This was first reported by me in 1981 [24]. A detailed current account of PION and its management is available in a recent publication based on my research [60].
AION is of two types [29, 36, 49, 74], depending on what causes it:
Arteritic AION: This is the most serious type and is due to a disease called giant cell arteritis or temporal arteritis.
Non-arteritic AION:: This is the usual, most common type, with many different causes but not associated with giant cell arteritis.
IT IS EXTREMELY IMPORTANT TO KNOW THAT THE CORRECT TREATMENT FOR AION DEPENDS ON WHETHER IT IS THE ARTERITIC OR THE NON-ARTERITIC TYPE.
To make a correct diagnosis of AION and its type, the doctor needs to find out all the required information. He/she might ask the following questions, because painless, loss of vision due to AION may be the first symptom of, or a complication of other diseases.
Giant cell arteritis is the prime medical emergency in ophthalmology because of its dreaded complication of visual loss in one or both eyes, which is preventable if these patients are diagnosed early and treated immediately and aggressively with systemic corticosteroids. This is a disease of persons aged 50 years or older - more common among women than men [40]. About 80% of patients with AION due to giant cell arteritis will have felt generally unwell for some time [40, 42]. However, 20% have no such general symptoms and are perfectly healthy [42]. Some patients may have a feeling of "euphoria," or well-being, so that they are quite unconcerned about their loss of vision and even insist they can see quite well when it is clear that they can't. Patients feeling like this may want to refuse treatment aimed at stopping the progress of giant cell arteritis and blindness, so it is important to have the cooperation and understanding of all family members. To help determine if giant cell arteritis is present, a physician will want to know about the following problems:
Visual acuity testing: The ophthalmologist needs to know at every visit just how much the patient can see, so that changes in the amount of affected vision can be determined. In addition to reading the eye charts for distance and near visual acuity, patients will often have visual field testing.
Visual field testing: A visual field test measures both central (straight ahead) and peripheral (side) vision. A defect in the visual field test shows which optic nerve fibers are affected by the disease An eye with normal 20/20 (or 6/6) visual acuity can have fairly marked visual field loss in the periphery, so a normal visual acuity does not guarantee that there is no visual loss.
Relative afferent pupillary defect: The pupil, located in the center of the iris, is the part of the eye which gets larger and smaller according to the amount of light. The optic nerve plays an important role in this reaction to light, so if the pupil is constricting to light less than normal it shows that there is damage to the optic nerve or the retina. The pupils are tested by shining a light into each eye, causing the pupils to constrict. As the light is moved from one eye to the other, the pupils should constrict to about the same size. If they do not, a relative afferent pupillary defect is present in the involved eye.
Tonometry (glaucoma testing): The pressure in the eye (intraocular pressure) is measured with an instrument called a tonometer. This simple, painless procedure is essential to obtain full information about the circulation in the eye. If there is any doubt about whether the pressure is high the patient may need to be tested several times over a whole day, because intraocular pressure can vary at different times of the day. A person may not know he/she has high pressure in the eye until after visual loss has occurred.
Fundus photograph and fluorescein fundus angiography: Photographs of the optic disc and the back of the eye may be taken on several occasions during the course of the disease to find out the amount of swelling of the optic disc and its resolution. Fluorescein fundus angiography is a routine dye test, done to find out the state of circulation in the back of the eye and optic disc.
More about fluorescein angiography
Blood tests and referrals to specialists in other areas: Blood tests which will be done immediately are the Erythrocyte Sedimentation Rate (ESR) and C-reactive protein (CRP). The results of these tests are available within a couple of hours. These two blood tests are extremely important to find out if a patient has giant cell arteritis. Both are usually abnormally high in giant cell arteritis.
Many other blood studies may be needed to find out if there is anything else wrong with the patient, and may help in finding the reason for the development of AION, such as diabetes or collagen vascular disease. Cholesterol and/or triglyceride levels will be checked because high levels lead to "hardening of the arteries." The results of these blood studies may indicate a need for follow-up by a local physician or referral to a specialist in hematology.
Blood pressure will be taken to determine if the patient has high blood pressure (hypertension). A cardiologist or cardiovascular specialist may be consulted if it seems that the cause of AION is a blood clot from somewhere else in the body, or that the heart, carotid arteries, or generalized "hardening of the arteries" may be contributing to the ischemia in the eye. Our recent studies have shown that abnormal fall of blood pressure during sleep is a serious risk factor for AION in the vast majority [12, 14, 15, 52, 60]. This can be tested by recording the blood pressure every 10 to 20 minutes over a 24 hour period, with an ambulatory blood pressure monitor
Temporal artery biopsy: When the patient's symptoms, the eye examination, elevated ESR and CRP, and fluorescein angiography make it seem likely that he/she has giant cell arteritis, a temporal artery biopsy will be done. The temporal artery lies just under the skin on the side of the forehead, or temple. The area is anesthetized and a small cut made in the skin to expose the artery. About an inch of the artery is removed for examination and the area is sutured with several stitches (which will be removed in about a week). This sample of the artery is examined under a microscope by a pathologist to determine whether there is inflammation of the artery. It may take several days before this result is known; however, treatment for giant cell arteritis may be started immediately if there is a strong suspicion of giant cell arteritis, even before the biopsy is done, because of the very high risk of blindness if adequate treatment is not given soon enough. Starting treatment before the biopsy is done does not interfere with the results.
Recent books by Dr. Sohan Singh Hayreh
Hayreh SS (2011). Ischemic Optic Neuropathies: Springer. 456 pages. ISBN: 978-3642118494
Hayreh SS (2015). Ocular Vascular Occlusive Disorders: Springer. 851 pages. ISBN: 978-3319127804