Chief Complaint: Redness and irritation in both eyes
History of the Present Illness: A 78-year-old male was referred by an outside ophthalmologist for evaluation of chronic irritation and dry eye unresponsive to aggressive lubrication, punctal occlusion, and Blephamide® (prednisolone/sulfacetamide) drops. In addition to dry eye symptoms, he had noticed a gradual decrease in vision in his right eye.
Past Ocular History
Past Medical and Surgical History
Medications
Family History: The patient’s mother has cataracts.
Social History: He occasionally drinks alcohol and denies use of tobacco or drugs.
Review of Systems: Unremarkable other than ocular symptoms.
Visual acuity (without correction):
Pupils: No RAPD OU
Extraocular motility: Full OU
Confrontational visual fields: No deficits OU
Intraocular pressure
External: Moderate diffuse facial erythema with telangiectasias and scattered papules on the forehead, cheeks, and nose.
Lids: Normal OU
Conjunctiva/Sclera: Atypical nasal pterygium OD at 3 o’clock with extension to paracentral cornea, no injection OU
Cornea: Whorled haze OD with limbal stem cell deficiency from 7 to 5 o’clock, clear central cornea OS with limbal stem cell deficiency from 9 to 1 o’clock; conjunctivalization of cornea with injection near limbus OD>OS
Anterior chamber: Deep and quiet OU
Lens: Posterior chamber intraocular lens OU
Vitreous: Normal OU
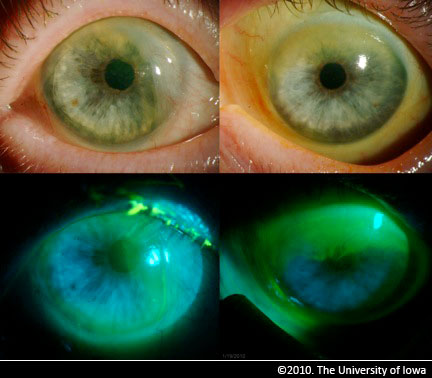 |
On initial presentation, the patient’s symptoms and findings appeared most consistent with severe ocular surface dysfunction resulting from bilateral limbal stem cell deficiency right greater than left. There was no obvious etiology for the stem cell deficiency and it was initially thought to be secondary to ocular rosacea. The patient did not improve after a trial of aggressive lubrication with preservative-free artificial tears and anti-inflammatory therapy including topical loteprednol and cyclosporine as well as doxycycline by mouth.
One month following his initial presentation, his best-corrected visual acuity had dropped to 20/250 OD with whorled corneal haze due to stem cell deficiency. He underwent pterygium excision OD with ocular surface reconstruction including amniotic membrane transplant (AMT). Due to an atypical appearance of the pterygium and underlying limbus intraoperatively, there was some suspicion for ocular cicatricial pemphigoid (OCP). The pterygium was sent to pathology for immunofluorescence studies including IgG, C3, and fibrinogen. Analysis of the specimen showed faint linear staining for IgG and C3 in the epithelial basement membrane zone with stronger staining for fibrinogen consistent with OCP (see figure 6).
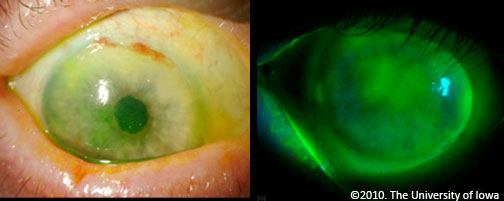 |
Postoperatively, the patient’s ocular surface was optimized with a bandage soft contact lens, autologous serum tears, antibiotic prophylaxis, and punctal occlusion. Topical and oral anti-inflammatory therapy was continued with the addition of mycophenolate mofetil (Cellcept®) 1000mg BID after pathologic confirmation of OCP. Seven months after AMT the patient’s vision had improved to 20/30 OD with marked improvement of corneal pannus and haze despite persistent stem cell deficiency. There has been no progression OS to date, and vision remains 20/20.
Diagnosis: Ocular cicatricial pemphigoid
Differential Diagnosis: ocular rosacea, limbal stem cell deficiency, pterygium, atopic keratoconjunctivitis, sarcoidosis (rare)
Discussion:
Ocular cicatricial pemphigoid (OCP) is a chronic, progressive, potentially blinding systemic autoimmune disease thought to be due to a type II (antibody dependent) hypersensitivity response against the hemidesmosome-epithelial membrane complex in conjunctival basement membrane (Muna, 2005).
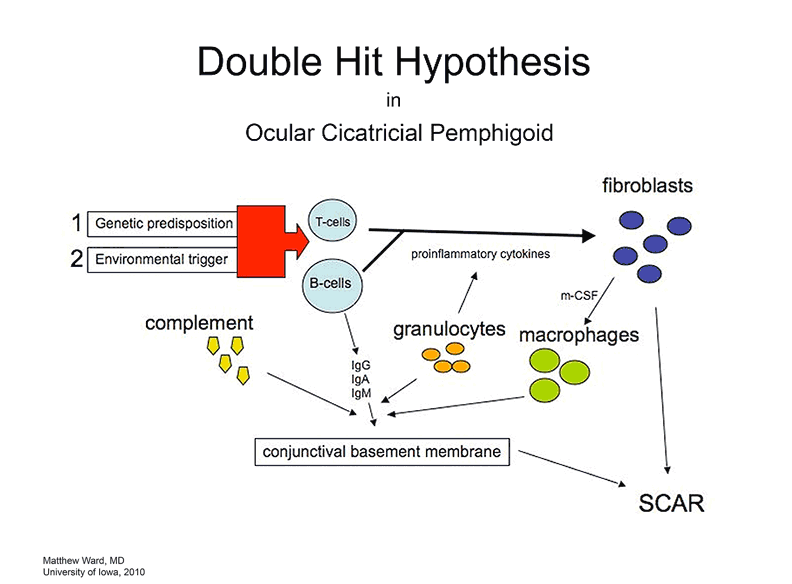
A "two hit" hypothesis has been proposed to explain the etiology of OCP (Razzaque, 2001). It is thought patients with an underlying genetic predisposition (first hit) sustain a second disease-causing hit by an unknown environmental factor which precipitates immunologic disarray in mucosal tissue (see figure 4).Specific human leukocyte antigen (HLA) associations have been described including DR-2, DR-4, DQw7, HLA A2, HLA B*8, HLA B*49 , HLA B*35. Onset of OCP has been observed after use of drugs as systemic practolol, topical pilocarpine, epinephrine, phospholine iodide, and timolol (Muna, 2005).
 |
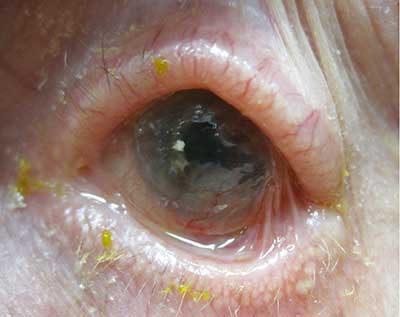
The classic ocular lesions are conjunctival shrinkage, fornix foreshortening, and symblepharon, though as this case illustrates, these are not necessary findings. The diversity of OCP presentation represents a significant diagnostic challenge—especially when early disease mimics some of the most frequently encountered and relatively benign ocular complaints.
 |
The disease can occur without ocular involvement resulting in ulceration and scarring at other sites including the pharynx, larynx, trachea, urethra, esophagus, vagina, or anus. In these cases it is simply termed "mucous membrane pemphigoid." Mouth ulcers are common (75-100% of cases) and can be a helpful diagnostic clue—especially in early stages of ocular involvement (Foster, 2008). Blistering skin involvement is present in up to 25% of OCP (ibid).
The gold standard for diagnosis is conjunctival biopsy with direct immunofluorescence testing for IgG, IgA, IgM, and C3. Positive staining reveals a linear pattern in the basement membrane zone of the conjunctival epithelium similar to that seen in other anti-basement-membrane-antibody conditions such as Goodpasture disease of the kidney (see figure 6).
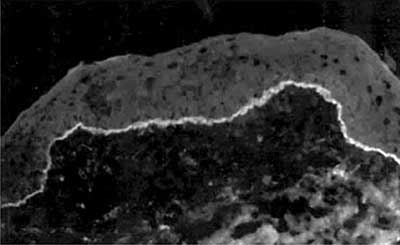 |
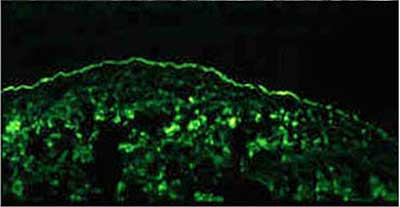 |
Progression of disease is measured in months and years and, without therapy, results in keratinization of the ocular surface and cicatricial ankyloblepharon (fusion of palpebral and bulbar conjunctiva, not to be confused with congenital palpebral ankyloblepharon, which describes a localized adhesion of the upper and lower eyelids). Though there are reports of spontaneous remission of OCP, these cases are the exception.
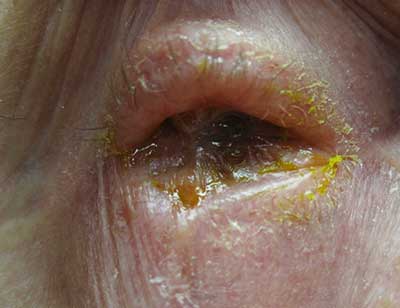 |
Foster and Muna suggest a staging system to aid in guiding management (Muna, 2005):
| Stage 1 conjunctivitis, subepithelial fibrosis | Stage 3 symblepharon |
| Stage 2 conjunctival shrinkage fornix foreshortening | Stage 4 ocular surface keratinization cicatricial ankyloblepharon |
True incidence of the disease is difficult to quantify because the diagnosis is not usually made until significant progression has occurred. It has been suggested that most cases are not diagnosed until stage 3.
OCP presents many challenges both in diagnosis and management. It is important to keep cicatricial pemphigoid in mind for patients with extended or recurrent conjunctivitis—especially if any signs of fibrosis are observed. Once the diagnosis is made, proper management must address both the pathophysiology of the disease and ocular surface dysfunction.
In terms of medical therapy, chronic steroid-sparing immunomodulators are the mainstay. Dapsone is a common initial agent, but requires initial testing to rule out hemolytic anemia, agranulocytosis, hepatitis, and G-6-PD deficiency. Agents such as methotrexate, azathioprine and mycophenolate mofetil have also been used. Recent evidence suggests that monotherapy with mycophenolate mofetil controls OCP in up to 70% of patients within one year of presentation with fewer side effects than other agents (Thorne, 2010). This evidence was corroborated by the outcome of the present case. Oral corticosteroids are effective at controlling inflammation, but symptoms usually recur once they are tapered. Ahmed and colleagues recommend reserving them only for the most severe inflammation and tapering down within the first 3 months (Ahmed, 2004). Targeted therapy with monoclonal antibodies (daclizumab and rituximab) has also shown some promise and was recently shown to arrest recalcitrant OCP when used in combination with intravenous immunoglobulin (IVIG) (Papaliodis, 2003; Foster, 2010). When initiating steroid-sparing immunomodulatory therapy, it is reasonable to enlist the help of an internist or rheumatologist to ensure appropriate monitoring. Aggressive lubrication is of utmost importance to optimize the ocular surface—preferably with preservative-free artificial tears and ointment or autologous serum tears if needed. Doxycycline, topical corticosteroids, and cyclosporine can help to reduce inflammation and increase tear production.
Surgical therapy may be indicated when scarring becomes advanced and/or ocular surface decompensation threatens permanent visual decline. Mucous membrane grafting from sites such as the nose and hard palate can slow or relieve cicatrization and treat symblepharon. In the present case, AMT was indicated to address conjunctivalization of the cornea and proved effective at reducing corneal neovascularization and alleviating discomfort. However, AMT did not improve the stem cell deficiency and the possibility remains that this patient will require keratolimbal allograft or a keratoprosthesis. Medical therapy after AMT should include a bandage soft contact lens to provide a smooth precorneal surface and protect against corneal breakdown and melt. Medroxyprogesterone (MPG) can be added to reduce the stimulus for neovascularization.
In stage 4 OCP, the type II Boston keratoprosthesis may be the only means of preserving or restoring visual function. This device creates a clear aperture through the upper eyelid (see figure 8). The lids are permanently sutured closed resulting in total coverage of the ocular surface.
EPIDEMIOLOGY:
|
SIGNS:
|
SYMPTOMS:Ocular
Systemic
|
TREATMENT:
|
Muna A, Foster CS. Ocular Cicatricial Pemphigoid in Uveitis and immunological disorders. Editors: Pleyer, U., Mondino, B., Berlin: Springer-Verlag; 2005.
Papaliodis GN, Chu D, Foster CS. Treatment of ocular inflammatory disorders with daclizumab. Ophthalmology 2003 Apr;110(4):786-789. http://www.ncbi.nlm.nih.gov/pubmed/12689903
Razzaque MS, Foster CS, Ahmed AR. Tissue and molecular events in human conjunctival scarring in ocular cicatricial pemphigoid. Histol. Histopathol 2001 Oct;16(4):1203-1212. http://www.ncbi.nlm.nih.gov/pubmed/11642740
Razzaque MS, Foster CS, Ahmed AR. Tissue and molecular events in human conjunctival scarring in ocular cicatricial pemphigoid. Histol. Histopathol 2001 Oct;16(4):1203-1212. http://www.ncbi.nlm.nih.gov/pubmed/11642740
Razzaque MS, Foster CS, Ahmed AR. Tissue and molecular events in human conjunctival scarring in ocular cicatricial pemphigoid. Histol. Histopathol 2001 Oct;16(4):1203-1212. http://www.ncbi.nlm.nih.gov/pubmed/11642740
Razzaque MS, Foster CS, Ahmed AR. Role of macrophage migration inhibitory factor in conjunctival pathology in ocular cicatricial pemphigoid. Invest. Ophthalmol. Vis. Sci 2004 Apr;45(4):1174-1181. http://www.ncbi.nlm.nih.gov/pubmed/15037585
Razzaque MS, Foster CS, Ahmed AR. Role of enhanced expression of m-CSF in conjunctiva affected by cicatricial pemphigoid. Invest. Ophthalmol. Vis. Sci 2002 Sep;43(9):2977-2983. http://www.ncbi.nlm.nih.gov/pubmed/12202518
Ahmed M, Zein G, Khawaja F, Foster CS. Ocular cicatricial pemphigoid: pathogenesis, diagnosis and treatment. Prog Retin Eye Res 2004 Nov;23(6):579-592. http://www.ncbi.nlm.nih.gov/pubmed/15388075
Daniel E, Thorne JE, Newcomb CW, Pujari SS, Kaçmaz RO, Levy-Clarke GA, Nussenblatt RB, Rosenbaum JT, Suhler EB, Foster CS, Jabs DA, Kempen JH. Mycophenolate mofetil for ocular inflammation. Am. J. Ophthalmol 2010 Mar;149(3):423-432.e1-2.http://www.ncbi.nlm.nih.gov/pubmed/20042178
Wolf R, Matz H, Orion E, Tuzun B, Tuzun Y. Dapsone. Dermatol. Online J 2002 Jun;8(1):2.[cited 2010 Aug 7 ] http://www.ncbi.nlm.nih.gov/pubmed/12165212
Foster CS, Chang PY, Ahmed AR. Combination of rituximab and intravenous immunoglobulin for recalcitrant ocular cicatricial pemphigoid: a preliminary report. Ophthalmology 2010 May;117(5):861-869. http://www.ncbi.nlm.nih.gov/pubmed/20045562
Foster CS. Cicatricial Pemphigoid: eMedicine Ophthalmology. 2008 Dec;[cited 2010 Aug 7 ] Available from: http://emedicine.medscape.com/article/1191261-overview
Ward MS, Syed NA, Goins KM. Ocular Cicatricial Pemphigoid: atypical presentation as pseudopterygium and limbal stem cell deficiency. Eyerounds.org. September 27, 2010; Available from: http://www.EyeRounds.org/cases/122-Limbal-OCP.htm.

Ophthalmic Atlas Images by EyeRounds.org, The University of Iowa are licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported License.