
The University of Iowa
Department of Ophthalmology and Visual Sciences
September 27, 2017
Glaucoma is the leading cause of irreversible blindness in the world with an estimated global burden of over 64 million people that is projected to increase to 111.8 million by 2040.[1] Currently, the only modifiable risk factor for glaucoma is intraocular pressure (IOP), and lowering IOP is the mainstay of treatment to date. Traditionally, topical medications, laser, and incisional surgery (trabeculectomy or tube shunts) have been the primary means to lower IOP and treat glaucoma. Topical medications require patient adherence, and cost may present a barrier to access. Selective laser trabeculoplasty (SLT) is typically effective initially, but its effectiveness can decrease over time.[2] Trabeculectomy and tube shunt surgeries are highly effective but carry a higher risk of complications. In the Tube Versus Trabeculectomy (TVT) Study, IOP decreased by a mean of 41.4% with tube surgery and 49.5% with trabeculectomy with decreased requirement for medication in both groups at 5 years follow up.[3] However, early complications (≤ 1 month) were seen in 21% of tube group and 37% of the trabeculectomy group, and late complications (> 1 month) were seen in 34% of the tube group and 36% of the trabeculectomy group. The rate of reoperation for complications was 22% in the tube group and 18% in the trabeculectomy group.[4] As an alternative to traditional glaucoma treatments, minimally invasive glaucoma surgery (MIGS) has shown promise for the future management of glaucoma.
The cardinal features of MIGS, as proposed by Saheb and Ahmed in 2012 [5], are:
MIGS typically require shorter operation time and allow for more rapid recovery. MIGS can be combined with cataract extraction (CE) for patients with mild to moderate glaucoma and cataracts. While MIGS have been approved for and studied in open angle glaucoma (OAG), they have also been used for other forms of glaucoma, including pseudoexfoliation, post-traumatic, pediatric, and ocular hypertension. With a lower risk profile, MIGS may be well-suited as a first line surgical intervention for monocular or high-risk patients. Though early studies show MIGS may be less effective in lowering IOP than traditional glaucoma surgeries, MIGS do fill a gap in the treatment of patients who would benefit from lower IOP but do not warrant the risk of traditional surgery. The growing array of MIGS work by increasing trabecular outflow, increasing uveoscleral outflow, increasing subconjunctival outflow, or decreasing aqueous production.
| MIGS: Increase Trabecular Outflow |
Specifics / Procedure: |
|---|---|
| iStent Micro-Bypass | Heparin-coated, non-ferromagnetic titanium stent; 1.0 mm x 0.3 mm. Ab interno insertion into Schlemm's canal |
| Gonioscopy-assisted transluminal trabeculotomy (GATT) | Ab interno trabeculotomy using illuminated microcatheter (iTrack; Ellex) or prolene/nylon suture passed through a 1-2 mm goniotomy into Schlemm's canal 360° and lysed through the trabecular tissue |
| Trabectome | Ab interno trabeculectomy using combination of electrocautery, irrigation and aspiration |
| TRAB 360 Trabeculotomy | Ab interno trabeculotomy using disposable, non-powered device from which a flexible nylon-like trabeculotome is advanced into Schlemm's canal for 180° and then lysed (x2 to perform up to 360° trabeculotomy) |
| Kahook Dual Blade | Ab interno trabeculotomy using a single use, tapered, stainless steel blade |
Ab interno canaloplasty |
Illuminated microcatheter (iTrack; Ellex) and viscosurgical device used to cannulate and viscodilate Schlemm's canal |
| Hydrus Microstent | Crescent-shaped scaffold (8-mm long) composed of nickel-titanium alloy, Ab interno insertion into Schlemm's canal |
| Increase Uveoscleral / Suprachoroidal/ Supraciliary Outflow | |
| CyPass Micro-Stent | Fenestrated micro-stent, composed of biocompatible, polyimide material (6.35 mm x 510 mm, 300 mm lumen) Ab interno insertion between anterior chamber/sclera and suprachoroidal space |
| iStent Supra | Heparin-coated stent (4mm long, 0.16-0.17mm lumen) composed of polyethersulfone (PES) with a titanium sleeve. Ab interno insertion between anterior chamber/sclera and suprachoroidal space |
| Increase Subconjunctival Outflow | |
| XEN Glaucoma Treatment System |
Tissue-conforming tube implant (6-mm long) composed of gelatin and glutaraldehyde material Ab interno insertion from the anterior chamber, through sclera into the subconjunctival space, bleb forming |
| InnFocus MicroShunt | Flexible microshunt (8.5 mm x 0.350 mm, 70 μm lumen) composed of SIBS (poly(styrene-block-isobutylene-block-styrene)) Ab-externo, subconjunctival (via peritomy) insertion through scleral needle tract into anterior chamber, connecting it to sub-Tenon's space, bleb forming |
| Decrease Aqueous Production | |
| Endocyclophotocoagulation | Ab interno cyclodestruction of ciliary body epithelium using continuous energy (810nm wavelength) |
Table 1: Overview of MIGS procedures and descriptions.
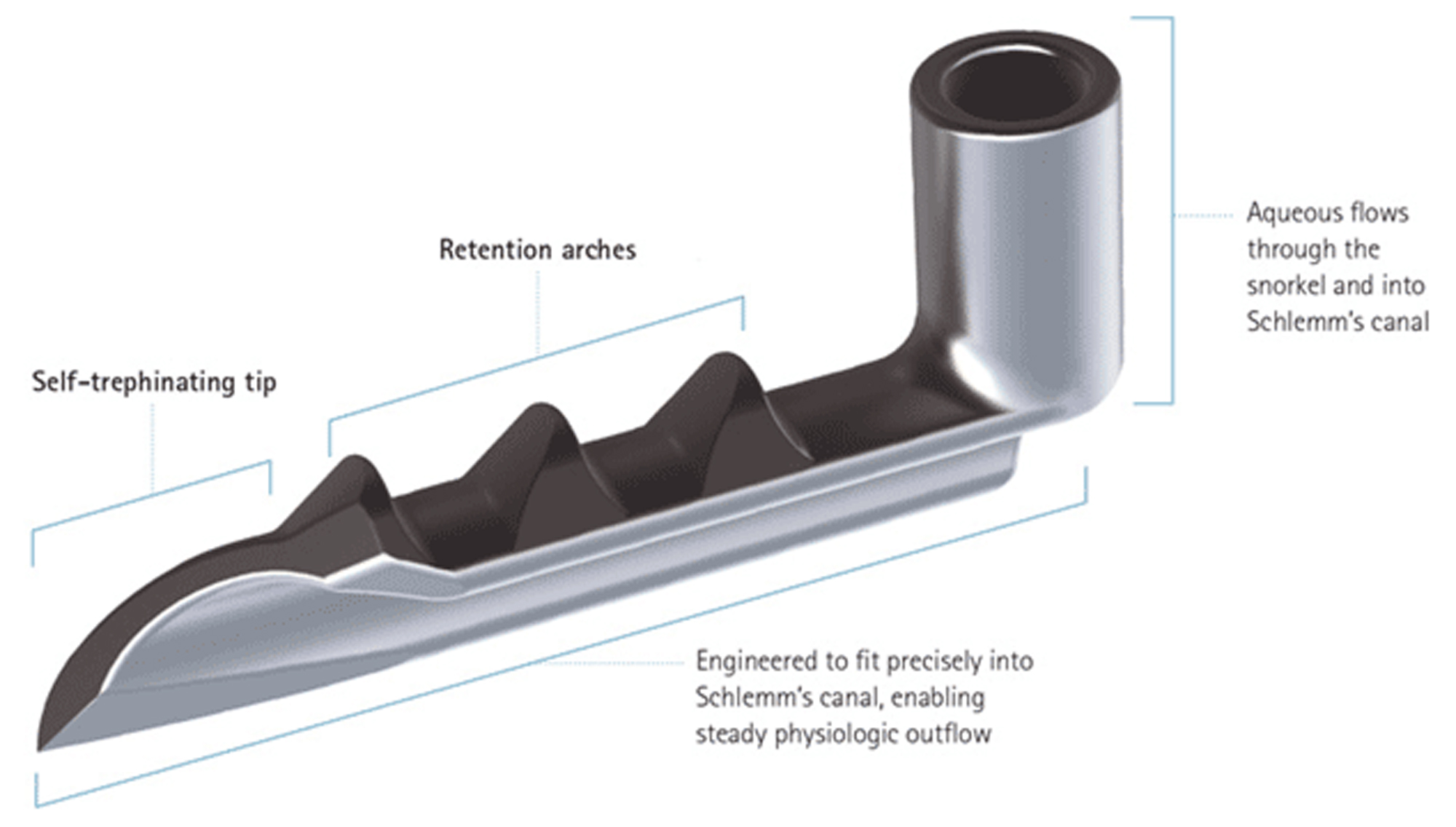
The iStent Trabecular Micro-Bypass Stent was the first FDA-approved (2012) trabecular microbypass. It works by incising then stenting Schlemm's canal to increase trabecular outflow. The iStent is a 1.0 mm x 0.3 mm, heparin-coated, non-ferromagnetic titanium (allowing magnetic resonance imaging up to 3 T). Its tapered design allows it to slide into Schlemm's canal, where it is then anchored by its 3 retention arches (Figure 1). The iStent is indicated for mild to moderate OAG previously treated with glaucoma drops, and performed in conjunction with CE.
![Figure 2. iStent Inject Source: Belovay et al. J Cataract Refract Surg. 2012;38(11):1911-7.[8]](MIGS-2-LRG.jpg)
Multiple randomized controlled trials (RCTs) have been conducted regarding the iStent. The iStent Study Group conducted the largest RCT including 240 subjects with mild to moderate glaucoma.
At 1 year, there was a significant difference in the iStent with CE group versus the control CE alone group in achieving the primary endpoint of an IOP ≤ 21 mmHg without glaucoma medications (72% vs 50%, p < 0.001). A secondary endpoint, decrease in IOP of ≥ 20% without glaucoma medications, was also significantly different between the 2 groups (66% vs 48%, p = 0.003). In addition, there was a significant difference in the reduction of medication burden among the groups at 1 year: 0.2 ± 0.6 medications were required in the iStent with CE group versus 0.4 ± 0.7 medications required in the CE alone group (p = 0.016).[6]
At 2 years, the proportion of subjects achieving primary outcome remained significantly higher in the iStent with CE group (61% vs 50%, p = 0.036), however the difference in IOP decrease ≥20% without glaucoma medications was not significantly different between the 2 groups (53% vs 44%, p = 0.09), and there was also no significant difference in number of glaucoma medications used (p = 0.09). After 2 years, IOP in the iStent with CE group was 8.4 mm Hg lower than baseline IOP, and IOP in the CE alone group was 7.5 mm Hg lower than baseline IOP, which was not a statistically significant difference.[7] The iStent Study Group showed that the iStent's efficacy was modest but allowed a prolonged reduction in IOP and medication burden. Importantly, there were no serious complications associated with the iStent. The most common complications were stent obstruction (4.3%) or malposition (2.6%), neither of which caused any adverse events.
To evaluate the efficacy of multiple iStents (2 or 3) in conjunction with CE, a prospective study of 53 eyes with OAG was conducted.[8] After 1 year, the overall mean IOP was significantly lower (14.3 ± 2.9 mmHg) than baseline (18.0 ± 4.0 mmHg) (p < 0.001) in each group (2 or 3) (p < 0.001). There was also a significant decrease in mean glaucoma medication at 1 year by 74% (2.7 ± 1.0 to 0.7 ± 1.1, p < 0.001). Additionally, 31 study eyes (59%) were off of all medications at 1 year. This study showed that multiple iStents can be safely implanted and result in effective IOP reduction on fewer glaucoma medications.
The iStent Inject is a 2nd generation iStent, which was developed with the premise that multiple iStents may be more effective than a single iStent. It is smaller in size (0.36 mm x 0.23 mm) and designed for perpendicular insertion into Schlemm's canal, no longer requiring parallel sliding of the iStent for positioning (Figure 2). Each device comes pre-loaded with 2 stents, so that both can be inserted in one procedure. A RCT of 192 subjects showed that implantation of 2 iStent Injects is comparable to medical treatment with two agents (latanoprost and timolol) in reducing IOP after 1 year.[9]Both the iStent Inject group and medication group had similar reductions in IOP (8.1 ± 2.6 mmHg and 7.3 ± 2.2 mmHg) and achieved similar rates of IOP reduction ≥ 20% compared to baseline at 1 year (94.7% and 91.8%). As with the original iStent, there was a highly favorable safety profile with the iStent Inject.[9]
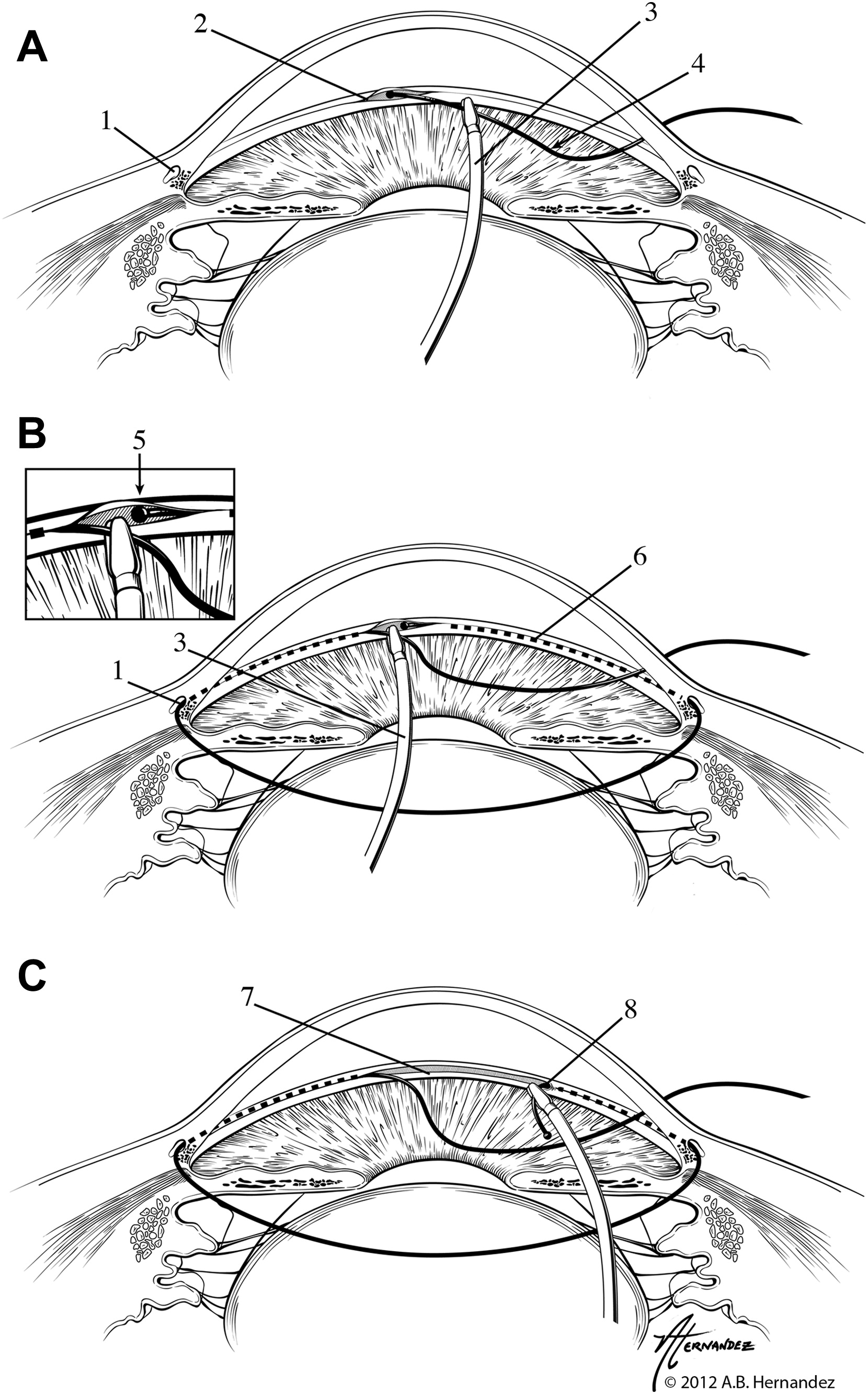
GATT was first described as a minimally-invasive form of ab interno trabeculotomy by Grover et al. in 2014.[10] An illuminated microcatheter (iTrack; Ellex) or suture (typically 5-0 or 6-0 nylon or prolene) is passed through a 1-2 clock hour goniotomy into Schlemm's canal then advanced for 360 degrees. It is then pulled centrally, applying force to lyse through the trabecular meshwork (TM) and creating a 360-degree trabeculotomy (fig. 3. [10]). The iTrack microcatheter has a 200-micron diameter shaft with a lubricated coating, along with a lighted tip, which can be constantly or intermittently illuminated to monitor catheter location. Like other MIGS, GATT is indicated for medically-uncontrolled OAG and can be performed with or without CE.
If video fails to load, use this link: https://youtu.be/y463tW3Ih0Q
Grover et al. conducted a retrospective review of 85 consecutive patients for whom GATT was performed with or without CE.[10] At 12 months follow-up of 57 primary OAG patients, IOP decreased by 11.1 ± 6.1 mmHg (40%) on an average of 1.1 ± 1.8 fewer glaucoma medications. For 28 patients with secondary OAG, IOP decreased by 19.9 ± 10.2 mmHg (57%) on an average of 1.9± 2.1 fewer medications. There was no statistically significant difference in IOP change related to lens status or whether concurrent CE was performed in eyes undergoing GATT.[10] The primary complication was transient hyphema in 30% of patients, which resolved in most patients by 1 month post-operatively. However, hyphema was still present in 3 patients at 1 month post-operatively, 1 patient at 3 months, and 1 patient at 6 months. Additional glaucoma surgery was needed in 9% (8/85) of the patients. Due to the nature of the procedure, there are several absolute contraindications: required anticoagulation, bleeding diatheses, angle closure, obscured angle structures, severe endothelial compromise, or intraocular lens instability. Relative contraindications include previous corneal transplant and an inability to elevate patient's head 30° during the first postoperative week.
In a retrospective review of the pediatric population, Grover et al. showed GATT to be a viable treatment option equivalent to ab externo trabeculotomy for the treatment of primary congenital glaucoma and juvenile OAG.[11]
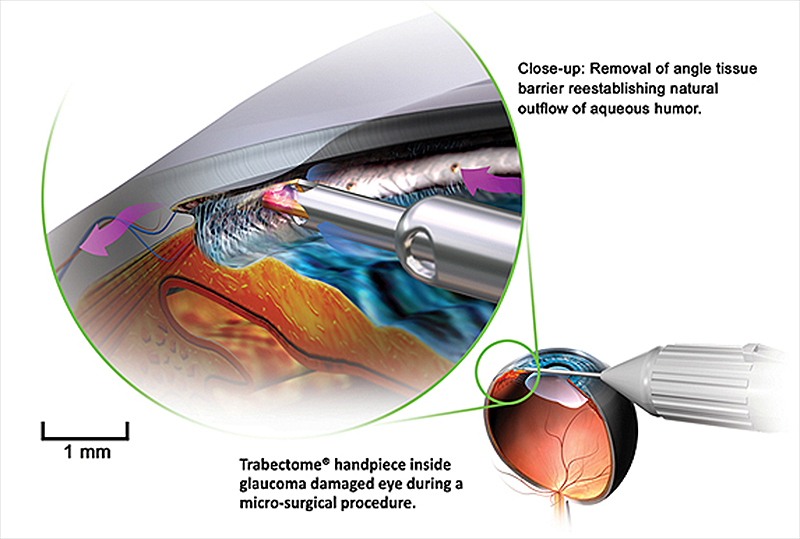
Trabectome is an FDA-approved (2004) device used to perform ab interno trabeculectomy (AIT). Trabectome combines electrocautery with irrigation and aspiration and consists of a 19.5 gauge handpiece with a bipolar 550 kHz electrode. The handpiece is disposable and requires a separate irrigation and aspiration console with a high frequency generator (Figure 4). As the electrocautery ablates trabecular meshwork up to 180 degrees, the natural drainage pathway is exposed. Simultaneously aspiration and irrigation are used to remove the ablated tissue to allow aqueous outflow. Initially, trabectome was indicated for primary OAG patients with uncontrolled IOP on maximal medical therapy, however many cases have reported the use of trabectome in a wide array of glaucoma subtypes, including narrow angle glaucoma.[12] In addition, AIT has been shown to be a good option in patients with pseudoexfoliation glaucoma and with a history of a failed trabeculectomy.[13 ,14]
No RCTs have been conducted examining trabectome. However, a meta-analysis compiled the data of 14 studies including 5,091 subjects and found an overall (66%) average rate of success, defined as IOP ≤ 21 mmHg with 20% decrease while avoiding reoperation, after 2 years.
The most common complications reported after trabectome were transient hyphema, peripheral anterior synechiae, corneal injury, and transient IOP spike. The rate of serious vision-threatening complications was minimal (< 1%), and these included hypotony (IOP < 5mmHg) 1 month after surgery, cyclodialysis cleft, choroidal hemorrhage, and endophthalmitis.[15]
TRAB 360 is a disposable, non-powered device used to perform an ab interno 360° trabeculotomy. The TRAB 360 device consists of a cannula, from which a flexible nylon-like trabeculotome is advanced into Schlemm's canal for 180 degrees (Figure 5). After the trabeculotomy is created, the trabeculotome can be retracted once and then advanced into the remainder of Schlemm's canal in the opposite direction for up to a total of 360 degrees. Like other trabeculotomy procedures, TRAB 360 is indicated for open angle glaucoma when IOP is not optimized on medical management.
Initial results from a study of the TRAB 360 device were reported by Sarkisian et al. at the 2015 American Society of Cataract and Refractive Surgery (ASCRS) Annual Meeting.[16] Surgical success, defined as IOP between 6-21 mmHg, was achieved in 25 of 30 eyes (83%) with or without glaucoma medications at the final follow-up visit. Mean IOP decreased from 19.8 ± 6.4 mmHg to 13.5 ± 4.6 at final follow up. Glaucoma medication burden was also decreased from a mean of 1.1 ± 1.2 pre-operative glaucoma medications to a mean of 0.2 ± 0.5 medications at the final visit. Mean time of follow-up was 131.5 ± 101.6 days. Transient hyphema was the most frequent postoperative complication and resolved by 1 week postoperatively in all cases.

The Kahook Dual Blade is a single-use, tapered, stainless steel blade used to incise and remove a nasal strip of trabecular meshwork tissue (Figure 6). After the tissue is engaged by the tapered tip, it travels up the angled ramp, where it meets two parallel blades. As the blades are advanced, this results in a ribbon of TM tissue that is separated from Schlemm's canal. Afterwards, the strip of TM is removed from the anterior chamber. Through one incision, the TM may be removed for a total of up to 180 degrees. Unlike Trabectome, there is no need for additional machinery for electrocautery, and theoretically, there is no collateral thermal damage. Currently, there are no published clinical trials on the Kahook Dual Blade. In a preclinical study of human donor corneoscleral rims, the Kahook Dual Blade showed more complete excision of TM tissue on histologic analysis with minimal damage to adjacent tissue as compared to a microvitreoretinal blade and the Trabectome (NeoMedix Inc.). In subsequent human eye perfusion studies, all devices showed significant reduction in IOP.[17]

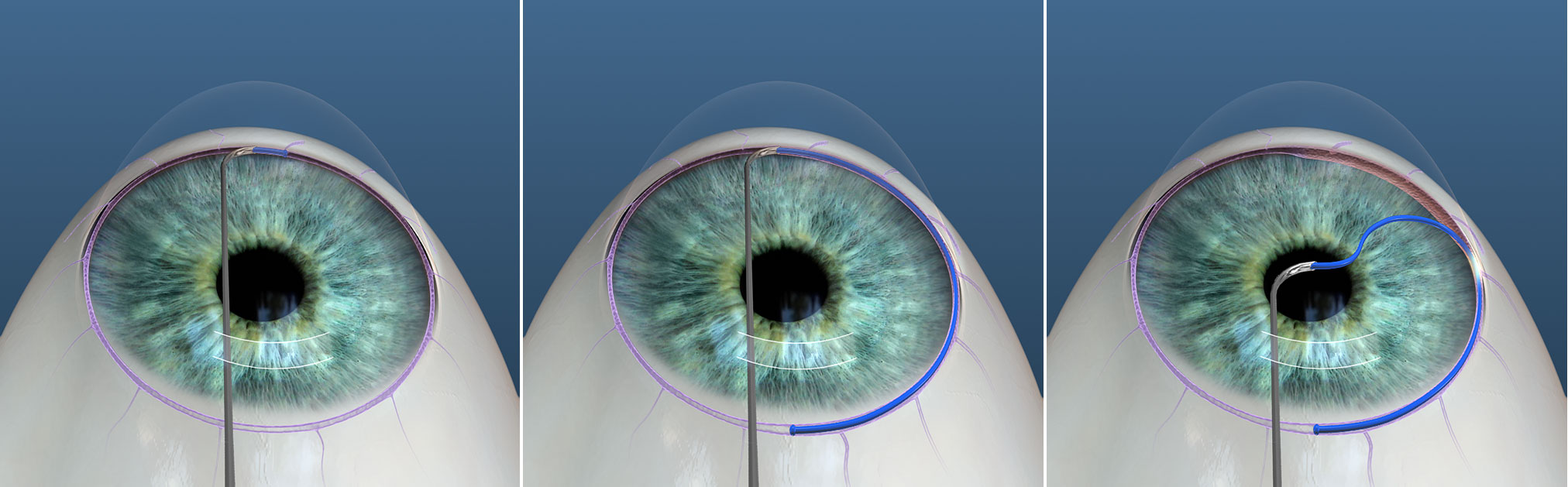
Ab interno canaloplasty (ABiC) increases aqueous outflow through cannulation of Schlemm's canal with an illuminated microcatheter (iTrack, Ellex), which is then withdrawn as an ophthalmic viscosurgical device is injected to viscodilate Schlemm's canal and the proximal collector channels. It has been theorized that viscodilation may also create microperforations within the TM to aid in aqueous outflow. As the viscoelastic is injected, blanching of episcleral vessels, which is indicative of a patent collecting system, serves as an indirect indicator of success. Indications for ABiC include mild to moderate OAG when maximal medical management and laser trabeculoplasty have failed. ABiC can be a better option for high risk monocular patients or for patients who are unable to stop anticoagulation, as ABiC minimally disrupts the TM with lower rates of hyphema. Contraindications to ab interno canaloplasty are similar to those of GATT (mentioned above), as proposed by Grover et al.[10] ABiC can be performed as a standalone procedure or in conjunction with CE.
In a retrospective review, Gallardo and Khaimi reported favorable results on 228 eyes treated with ABiC with and without CE.[18] Overall, mean preoperative IOP was 19.0 ± 6.5 mm Hg, and the mean preoperative number of glaucoma medications was 2.0 ± 1.0. At 12 months post-operatively, mean IOP decreased 30% to 13.3 ± 2.0 mm Hg, and the mean postoperative number of glaucoma medications decreased by 50% to 1.0 ± 1.0. The specific complications were not reported in Gallardo's and Khaimi's series, however in a case series of 20 eyes achieving similar success, there was only 1 reported complication of Descemet's detachment during injection of viscoelastic.[18] There were no reported cases of significant hyphema in either of these series.
If video fails to load, use this link: https://www.youtube.com/embed/mszdvAPw8Xw
VISCO360 Viscosurgical System is a device, similar to TRAB360, through which ABiC can be performed (Figure 7). There are no published data on the VISCO360. However, , there is an ongoing multicenter, RCT evaluating the safety and effectiveness of the VISCO 360 Canaloplasty in primary OAG as compared to SLT.[19]

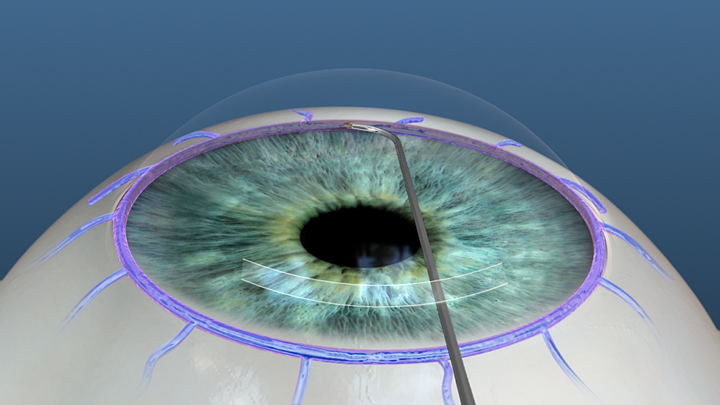
The Hydrus Microstent improves aqueous outflow into multiple collector channel ostia by serving as an "intracanalicular scaffold" for Schlemm's canal and a bypass of the TM. The Hydrus Microstent is an 8-mm ("3 clock hours") long, crescent-shaped scaffold composed of a nickel-titanium alloy (nitinol) with windows and spines (Figure 8) and comes preloaded in a hand-held injector. Hydrus implantation is currently being investigated in conjunction with CE and is indicated for mild to moderate OAG.
In a prospective, multicenter RCT (HYDRUS II), 100 OAG patients were randomized to a Hydrus Microstent with CE arm and a CE alone arm. At 2 years, there was a statistically significant difference in the primary endpoint of a 20% reduction in diurnal, washed-out IOP compared to baseline. The primary endpoint was met in 80% (40/50) of patients in the Hydrus Microstent with CE arm compared to 46% (23/50) in the CE alone arm (p = 0.001). At 2 years, the mean reduction in IOP in the Hydrus Microstent with CE arm (9.4 mmHg) was significantly lower than in the CE alone arm (7.4 mmHg) (p = 0.01). There was also a statistically significant difference in mean glaucoma medications per patient after 2 years, decreasing from 2.0 ± 1.0 to 0.5 ± 1.0 medications in the Hydrus Microstent with CE arm and from 2.0 ± 1.1 to 1.0 ± 1.0 in the CE alone arm (p = 0.02). The proportion of patients who did not require medications after 2 years was 73% in the Hydrus Microstent with CE arm and 38% in the CE alone arm (p = 0.001). In this study, there was a significantly higher rate of focal peripheral anterior synechiae (PAS) in the Hydrus Microstent with CE arm at 2-years of follow-up (6/50, p = 0.01). However, PAS had no effect on outcomes, as IOP and medication use were similar to that in the overall Hydrus Microstent with CE arm.[20] Other prospective, multicenter RTCs are being conducted including the HYDRUS III study, which compares the Hydrus Microstent with the iStent, and the HYDRUS IV study, which is the largest MIGS RCT, including 556 subjects with mild to moderate glaucoma.[21]

Update 9/27/2018
The CyPass Micro-stent was voluntarily withdrawn from the global market by Alcon on August 28, 2018 .[39] The COMPASS-XT study, evaluating the long-term safety of the CyPass Micro-stent, showed a statistically significant increase in endothelial cell loss in the CyPass group after 5 years of follow up. The American Society of Cataract and Refractive Surgery (ASCRS) established a task force to evaluate COMPASS-XT data and provide recommendations for managing patients with the CyPass Micro-stent device. The task force recommends that providers consider repositioning, removal, or trimming the CyPass device if there is clinically-significant evidence of corneal decompensation and >1 ring of the CyPass device is visible. If there is no clinically-significant evidence of corneal decompensation, then no intervention is recommended other than monitoring.[40]
The CyPass Micro-stent is a FDA-approved (2016) suprachoroidal shunt used to increase uveoscleral outflow. The CyPass device itself is a flexible, fenestrated micro-stent sized 6.35 mm x 510 μm with a 300 μm lumen and composed of biocompatible, polyimide material (Figure 9). It comes preloaded with the micro-stent on a guide-wire conformed to the shape of the sclera to facilitate dissection and insertion between the anterior chamber/sclera and suprachoroidal space. The CyPass Micro-stent is indicated for mild to moderate primary OAG.
The COMPASS Trial was a multicenter RCT including 505 subjects with mild to moderate primary OAG. Subjects were intraoperatively randomized into a CyPass Micro-stent with CE arm (374 subjects) and a CE alone arm (131 subjects).[22] At 2 years, there was a statistically significant difference in the primary endpoint, a 20% reduction in diurnal, washed-out IOP compared to baseline. This endpoint was met in 77% of the Cypass Micro-stent with CE arm compared to 60% in the CE arm (p = 0.001). After 2 years, the mean reduction in IOP in the Cypass Micro-stent with CE arm (7.4 mmHg) was significantly lower than in the CE arm (5.4 mmHg) (p < 0.001). There was also a statistically significant difference in mean glaucoma medications per patient and the proportion of patients who did not require medications after 2 years. At 2 years, patients in the Cypass Micro-stent group decreased the number of medications from a baseline of 1.4 ± 0.9 to 0.2 ± 0.6. In the CE alone arm, medication use decreased from a baseline of 1.3 ± 1.0 to 0.6 ± 0.8. After 2 years, 85% in the Cypass Micro-stent with CE arm and 59% in the CE alone arm did not require medications (p < 0.001). There were no significant differences in the rate of adverse events between the two arms. However, reported adverse events included transient BCVA loss ≥ 2 lines (8.8%), visual field loss progression (6.7%), transient iritis (8.6%), transient corneal edema (3.5%), and transient hypotony (2.9%).[22]

The iStent SUPRA is a suprachoroidal stent, which is also designed to increase uveoscleral outflow. The device is a heparin-coated, 4 mm tube with a 0.16 - 0.17 mm lumen, made of polyethersulfone (PES) with a titanium sleeve. As with the original iStent, the device has retention ridges to hold the device in place (Figure 10). The iStent SUPRA is preloaded into an injector device, which is used to direct the device between the anterior chamber/sclera and suprachoroidal space. Placement of the iStent SUPRA can be combined with CE and is indicated for mild to moderate OAG.
Katz et al. conducted a prospective study of 80 subjects with moderate to advanced OAG on 2 glaucoma medications. Eighty subjects received the iStent SUPRA and travoprost postoperatively and agreed to be followed for 5 years.[23,24] After follow-up through 1 year (n=42), 98% of eyes met the primary endpoint of 20% reduction in IOP with reduction in 1 medication. The mean medicated diurnal IOP decreased from 20.4 mmHg to 12.7 mmHg at 1 year and to 11.9 mmHg at 2 years. The preoperative mean washed-out IOP was 24.8 mmHg, and after a 1 month medication wash-out at both 1 and 2 year time points, mean washed-out IOP was 16.7 mmHg and 17.0 mmHg respectively. Additionally, an IOP ≤ 15 mmHg was achieved in 90% of eyes on travoprost at 2 years. Transient hypotony (IOP < 5mmHg) was observed at week one in 2 eyes, both of which resolved by 1 month. One case was associated with choroidal detachment, which resolved by 3 months. BCVA was stable or improved in all but 1 subject due to cataract progression. To gain FDA approval, Glaukos recently completed enrollment (February 2017) for a prospective RCT including 505 subjects with moderate to mild primary OAG and cataract.[25]
![Figure 10. A) iStent SUPRA device. B) iStent SUPRA inserted into suprachoroidal space. Source: Surgical Innovations in Glaucoma. Springer[26] Images used with permission from Springer-Verlag, New York](MIGS-10-LRG.png)
The XEN gel stent is an FDA-approved (2016) subconjunctival stent, which allows aqueous outflow from the anterior chamber into the subconjunctival space to lower IOP. It is a 6 mm tube composed of gelatin and glutaraldehyde. The implant is preloaded within a 27-gauge needle on a disposable injector. The device is inserted into the anterior chamber, passed ab interno, and then tunneled through sclera to deploy the device within the subconjunctival space (Figure 10). After implantation, the device creates a filtering bleb. It may be performed with or without adjuvant antimetabolites (mitomycin C), which in this case would be injected beneath the conjunctiva.
There are three sizes of the XEN gel stent based on lumen diameter. The XEN45 (45 μm lumen) is the only currently FDA-approved size and is for use in patients with refractory glaucoma failing surgical treatment, primary OAG, pseudoexfoliative, or pigmentary glaucoma with open angles that have failed maximum medical therapy.
In a multicenter, single-arm clinical trial including 65 patients, the implantation of the XEN45 gel stent resulted in a 20% reduction in mean diurnal IOP from baseline on the same or fewer number of glaucoma medications in 76.3% of subjects at 12 months.[27] There was a decrease in IOP from a mean baseline of 25.1 ± 3.7 mmHg to 15.9 ± 5.2 mmHg. At 12 months, the mean number of glaucoma medications decreased from baseline of 3.5 ± 1.0 to 1.7 ± 1.5. Most common adverse events included hypotony (IOP < 6 mmHg), BCVA loss ≥ 2 lines (n = 4 at 12 months), and an IOP increase > 10 mmHg from baseline. The reported cases of hypotony were transient and resolved in all cases without sequelae. Six of the 17 subjects with an IOP increase > 10 mmHg from baseline had a secondary glaucoma procedure performed prior to the 12-month visit. Needling procedures were needed in 21 subjects due to a flat bleb with the absence of microcysts, fibrotic or blocked bleb filtration area, or a high risk of bleb failure as determined by investigators.
Two other sizes of the XEN gel stents (63 μm and 140μm) have been studied in pilot trials, showing significant IOP reduction and achieving complete success in 29/34 (85%) patients and qualified success in 40/45 (89%) patients. However, there were high rates of eyes requiring needling with or without antifibrotics in both studies: 12/37 (32%) and 21/49 (47%) respectively. Though there were few complications, anterior chamber fill with a viscoelastic was required in 2/34 (6%) and 4/45 (9%) of subjects.[28 ,29] Antifibrotics were not used intraoperatively, but it is thought that the rate of needling would be lower if used during placement of the XEN gel stent, which is currently under study.
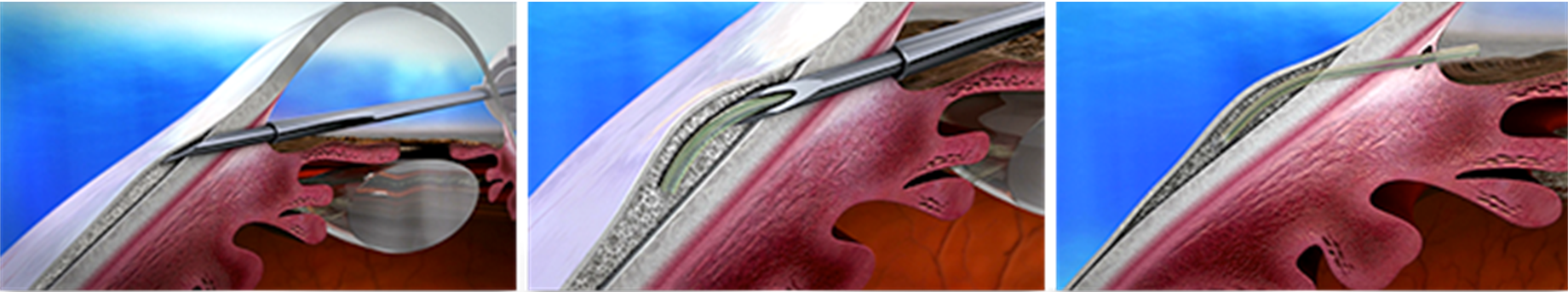
The InnFocus Microshunt also allows aqueous drainage into the subconjunctival space, however it is placed using an ab externo approach. The device is 8.5 mm x 0.350 mm with a 70 μm lumen and composed of SIBS [poly(styrene-block-isobutylene-block-styrene)] material which regulates aqueous flow (Figure 12). After making a small conjunctival peritomy, a needle is used to create a small scleral pocket, within which a smaller needle enters the anterior chamber. The device is then implanted and allows aqueous humor to drain from the anterior chamber into sub-Tenon's space to form a bleb. As in trabeculectomy, mitomycin C is routinely placed in the area of the intended bleb. InnFocus Microshunt implantation may be performed with or without CE. The InnFocus Microshunt is not yet FDA-approved, but is indicated for the treatment of mild, moderate, or severe OAG.
Few published studies have evaluated the InnFocus Microshunt, but clinical trials are being conducted to obtain FDA approval.[30] In a prospective, observational study of 23 eyes having failed maximal medical therapy, 14 eyes received the InnFocus Microshunt and 9 eyes received InnFocus Microshunt in conjunction with CE.[31] After 3 years of follow-up (n = 22), the qualified rate of success, defined as IOP ≤ 14 mm Hg and IOP reduction ≥ 20%, was 95%. IOP decreased from 23.8 ± 5.3 mmHg to 10.7 ± 3.5 mmHg, and the mean number of glaucoma medications decreased from 2.4 ± 0.9 to 0.7 ± 1.1. Reported complications included transient hypotony in 3 patients and transient choroidal effusion in 2 patients, all of which spontaneously resolved by week 3 and week 12, respectively. Additionally, 3 patients had iris-tube contact, 2 had hyphema, and 2 had exposed Tenon's capsule. No bleb leaks, infections, or serious long-term complications were reported.
![Figure 12. InnFocus Microshunt implanted between subconjunctival space and anterior chamber. Image used with permission from InnFocus, Inc.[30]](MIGS-12-LRG.jpg)
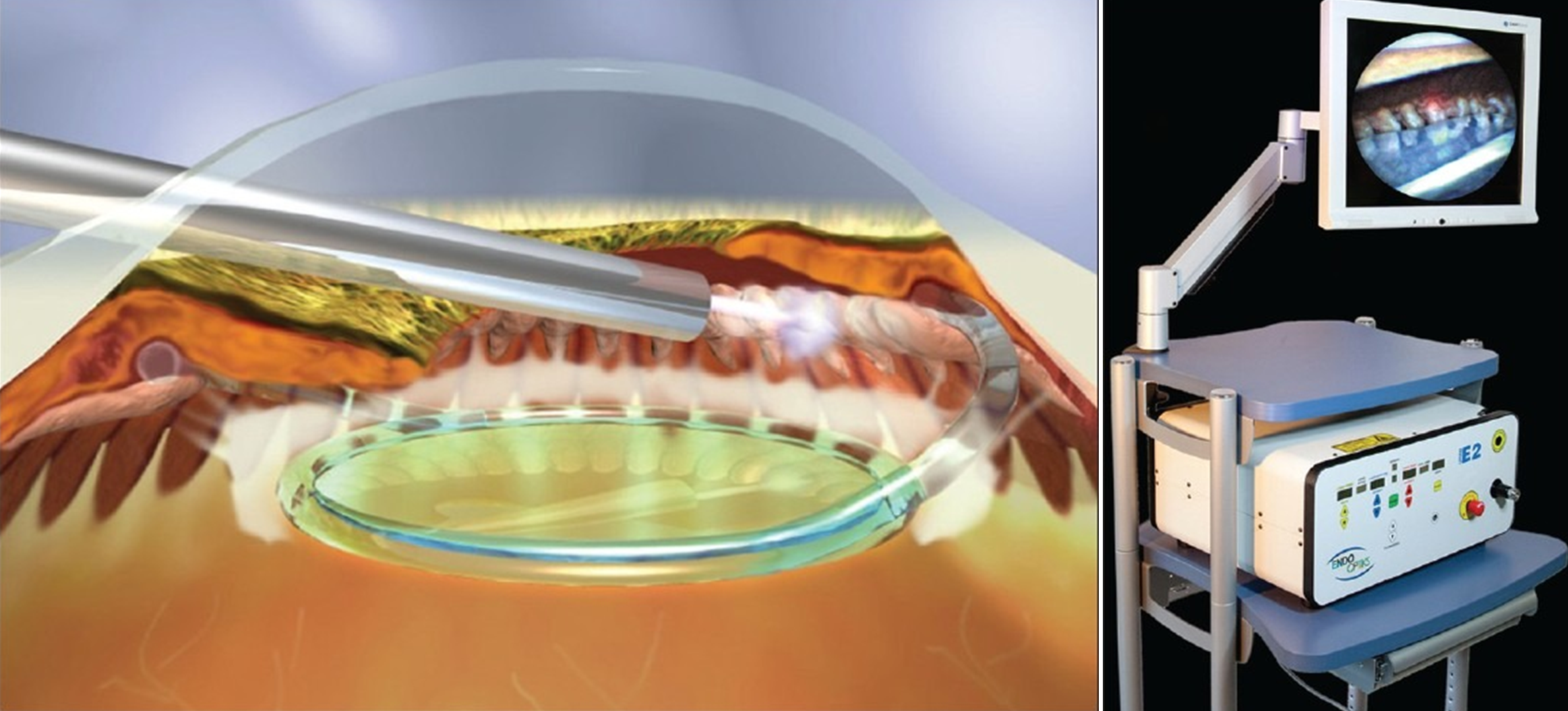
Endocyclophotocoagulation (ECP) consists of cyclodestruction of the ciliary body epithelium to reduce aqueous production and therefore IOP. The ECP probe is reusable device, which includes a laser source, camera, and light source. After it is introduced into the anterior chamber, the probe can then be directed towards the anterior ciliary processes to deliver precise continuous energy (810 nm wavelength) for successful photocoagulation, visualized as localized shrinkage and whitening of the processes (Figure 13). Through a single corneal incision, approximately 240 to 300 degrees of the ciliary processes can be treated, but incisions are needed for a 360-degree treatment. As expected, the greater the amount of processes treated, the greater the reduction in IOP and need for glaucoma medications.[32]
Excessive energy or overtreatment results in eruption of ciliary processes and may be accompanied by a "popping" sound. ECP induces an inflammatory response, and steroids (intravenous or oral) are often helpful to suppress inflammation and reduced IOP spikes. ECP may be utilized in many types of glaucoma (open or closed angle), including pediatric glaucoma.[33] ECP may be used as an initial management option for mild to moderate glaucoma or for patients with advanced glaucoma that have failed previous management.[34] ECP can be performed with or without CE but is better suited for pseudophakic or aphakic eyes, as ECP can lead to cataract and zonular damage.
In a recent, non-randomized prospective study, Francis et al. showed ECP with CE to be effective in decreasing IOP and medication burden, compared to CE alone in a group of 160 consecutive patients with medically-controlled OAG.[35] The rate of success, defined as IOP 5 - 21 mmHg and reduction in glaucoma medications without a rise in IOP, was significantly higher in the ECP with CE group compared to the CE alone group after 3 years (p < 0.01). Similarly, IOP and required glaucoma medications were significantly lower in the ECP with CE group as compared to the CE alone group. After 2 years, the mean IOP in the ECP with CE group (n = 80) decreased from baseline of 18.1 ± 3.0 mmHg to 16.0 ± 3.3 mmHg on fewer glaucoma medications (1.5 ± 0.8 to 0.4 ± 0.7), and the mean IOP decreased from 18.1 ± 3.0 mmHg to 17.3 ± 3.2 mmHg on fewer glaucoma medications (2.4 ± 1.0 to 2.0 ± 1.0) in the control group (n = 80). Reported adverse events included hyphema, inflammation, and IOP spike. Smaller, prospective studies have also shown ECP to be comparable to traditional incisional glaucoma surgeries with fewer complications.[36 ,37]
| MIGS Procedure | Decrease in IOP | Decrease in Medications | Study Type |
|---|---|---|---|
| iStent Micro-Bypass* [7] | 8.4 mmHg @ 2 years | 0.8 @ 2 years | Randomized controlled trial |
| iStent Inject [9] | 8.1 mmHg @ 1 year | Not available | Prospective, randomized trial |
| Gonioscopy-assisted transluminal trabeculotomy (GATT)* [10] | 8.4 mmHg @ 1 year | 1.9 @ 1 year | Retrospective review |
| Trabectome* [15] | 6.2 mmHg @ 2 years | 0.76 @ 2 years | Meta-analysis |
| TRAB 360 Trabeculotomy [16] | 6.3 mmHg @ 131.5 days** | 0.9 @ 131.5 days** | Retrospective review |
| Ab interno canaloplasty* [18] | 4.0 mmHg @ 1 year | 1.0 @ 1 year | Case-series review |
| Hydrus Microstent* [20] | 9.4 mmHg @ 2 years | 1.5 @ 2 years | Randomized controlled trial |
| CyPass Micro-Stent* [22] | 7.4 mmHg @ 2 years | 1.2 @ 2 years | Randomized controlled trial |
| iStent Supra [23 ,24] | 7.8 mmHg @ 2 years | Not available | Prospective, single arm clinical trial |
| XEN Glaucoma Treatment System [27] |
9.2 mmHg @ 1 year | 1.8 @ 1 year | Prospective, single arm clinical trial |
| InnFocus MicroShunt* [31] | 16.2 mmHg @ 3 years | 1.6 @ 3 years | Prospective, single arm clinical trial |
| Endocyclophotocoagulation* [35] | 2.1 mmHg @ 2 years | 1.1 @ 2 years | Prospective case-control study |
Table 2: Relative effectiveness of MIGS Procedures based on decrease in IOP and decrease in medications for selected studies. Note *Combined procedure MIGS with cataract extraction **Mean follow up.
One study conducted by Iordanous et al. compared the cost of Trabectome, iStent, and ECP to glaucoma medications in the Ontario Health Insurance Plan.[38] Though physician fees and start up costs were not included for each MIGS device, the cumulative cost savings of successful Trabectome, iStent, and ECP procedures as compared to mono-drug, bi-drug, and tri-drug therapy were shown to be modest over a 6 year period (Figure 14). Trabectome had the greatest cost savings, followed by ECP, and then iStent. While glaucoma management can be a financial burden, many other factors, such as effectiveness, safety, quality of life, or compliance, must be further considered as well. As MIGS are further developed and more widely used, more evidence and long-term follow-up will allow further study of cost-effectiveness.
| Cumulative Cost Per Patient (Canadian Dollar $, 2014) | ||||||
|---|---|---|---|---|---|---|
| 1 year | 2 year | 3 year | 4 year | 5 year | 6 year | |
| Trabectome | 744.00 | 744.00 | 744.00 | 744.00 | 744.00 | 744.00 |
| iStent | 1044.00 | 1044.00 | 1044.00 | 1044.00 | 1044.00 | 1044.00 |
| Endoscopic Photocoagulation | 244.00 | 244.00 | 244.00 | 244.00 | 244.00 | 244.00 |
| Medical Therapy | ||||||
| Mono-drug | 170.54 | 341.08 | 511.61 | 682.15 | 852.69 | 1023.23 |
| Bi-drug | 386.09 | 772.18 | 1158.28 | 1544.37 | 1930.46 | 2316.55 |
| Tri-drug | 528.12 | 1056.24 | 1584.36 | 2112.48 | 2640.60 | 3168.71 |
Table 3: Cumulative 6-Year Cost Comparison of Trabectome, iStent, and ECP versus Mono-drug, Bi-drug, and Tri-drug Therapy. Adapted from Journal of Glaucoma. 23(2):e112-e118, 2014.[38]
MIGS is a relatively new category of procedures with a limited number of studies showing largely acceptable safety profiles and modest efficacy. While MIGS surgeries currently appear unlikely to supplant traditional incisional glaucoma surgeries, they fill an important gap between medical therapy and incisional surgery for mild to moderate glaucoma and can often mitigate medication burden. As the area of MIGS continue to evolve, more rigorous studies with longer follow-up will increase our understanding of the full efficacy and safety of MIGS procedures.
DISCLAIMER: EyeRounds editorial board members, the authors, and the University of Iowa do not recommend or endorse any specific tests, drugs, devices, products, services, physicians, or medical institutions that may be mentioned or referenced on this web site. Manufacturers of products and procedures mentioned have no influence on the editorial content or presentation and such content does not imply or express endorsements of any product, service, or company.
Fox AR, Risma TB, Kam JP, Bettis DI. MIGS: Minimally Invasive Glaucoma Surgery. EyeRounds.org. posted September 27, 2017; Available from: https://eyerounds.org/tutorials/MIGS/