
The University of Iowa
Department of Ophthalmology and Visual Sciences
October 15, 2013
Retinal detachment is an important cause of decreased visual acuity and blindness. It is one of the most common ocular emergencies today, most frequently affecting the middle aged and elderly. It often requires urgent—and sometimes emergent—treatment. Because of the severity of this condition, it is an excellent topic for medical students to review.
The chance of developing a retinal detachment is related to a combination of risk factors and misfortune. The primary risk factors for retinal detachment include myopia, lattice degeneration, and advancing age.(1) Other risk factors are recent ocular surgery or trauma. For example, retinal detachment after cataract extraction ranges from 0.6 - 1.7% yearly.(2)
There is no specific way to decrease the incidence of retinal detachment, but understanding the symptoms of retinal detachment is important for all patients. The sooner patients can be seen when they have a retinal detachment, the better their visual prognosis will be.
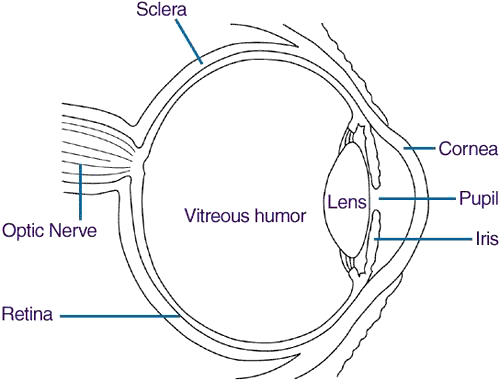
Think of the retina as the film of the camera. It is a thin layer that coats the internal border of the posterior eye. The retina is comprised of photoreceptors, neurons, and support cells. It is bordered anteriorly by the vitreous and posteriorly by the choroid. The central portion of the retina is the macula. The macula has a high concentration of photoreceptors and is responsible for capturing central vision. More specifically, the highest proportion of photoreceptors is in a central 1.5mm zone called the fovea. This area of high photoreceptor density is important in retinal detachment as its condition dictates the prognosis of the disease.
Internal and anterior to the retina is the vitreous. Think of the vitreous as clear jelly inside the eye comprised of type II collagen and hyaluronic acid. It comprises 80% of the volume of the eye and is responsible for providing mechanical support for our eyes during development.(3) However, there is little evidence of a functional purpose of the vitreous after our eyes have fully developed. Also, the vitreous breaks down and liquefies as we age.
External and posterior to the retina are the retinal pigment epithelium (RPE) and the choroid. The retinal pigment epithelium is between the retina and the choroid. It is responsible for nourishing the retina. It recycles vitamin A for use by the retina in the visual cycle, and it keeps fluid from accumulating underneath the retina. The choroid is a vascular layer immediately posterior to the RPE which provides blood to the outer layers of the retina (the inner layers of retina supplied by branches of the central retinal artery).
There are many classification schemes for retinal detachment. However, in the simplest terminology, there are three primary forms of retinal detachment. These include rhegmatogenous retinal detachments, tractional retinal detachments, and exudative retinal detachments.
Rhegmatogenous retinal detachment (RRD) is the most common form of retinal detachment. It affects approximately 0.007-0.018% of the population yearly.(4) Rhegma means “break” in Greek, and these retinal detachments are due to full-thickness breaks in the retina. These full-thickness breaks, most commonly due to and held open by vitreoretinal traction, allow fluid from the liquified vitreous to move under the neurosensory retina and further separate it from the retinal pigment epithelium.
Tractional retinal detachment occurs when vitreous membranes pull on the retina separating it from the RPE. No tears or holes are present in this type of retinal detachment. Vitreous membranes are usually gliotic, fibrous, vascular, or a combination. They commonly grow in response to proliferative vitreoretinopathies, most notably diabetic retinopathy.(5)
Exudative retinal detachment occurs due to an underlying condition causing buildup of fluid (exudate) between the neurosensory retina (photoreceptor layer) and the retinal pigment epithelium (RPE). Inflammatory conditions such as uveitis are the most common conditions responsible for this type of retinal detachment. Other rare but more serious conditions such as choroidal tumors can be responsible for exudative retinal detachments too.
he primary symptoms experienced during a retinal detachment are the sudden appearance of many floaters, flashes of light (photopsias), and/or a dark curtain over a portion of the visual field. Retinal detachments are almost always painless.
Every patient encounter should begin with a history of present illness from the patient. When a possible detachment is suspected, the presence or absence of the above symptoms should be documented. Ask about history of prior retinal detachment, retinal tears, and any prior ocular procedures. Identification of diagnosed ocular diseases and family history of retinal detachment are also very important in the proper workup of these patients.
Examination should start similar to other patients that present to the ophthalmology clinic. Visual acuity, intraocular pressure, visual fields, presence or absence of a relative afferent pupillary defect (RAPD), and extraocular motility should all be obtained. After these have been performed, the patient should have their pupils dilated for a thorough fundus exam. Examining both eyes is important as asymptomatic retinal pathology be identified in the fellow eye, too.
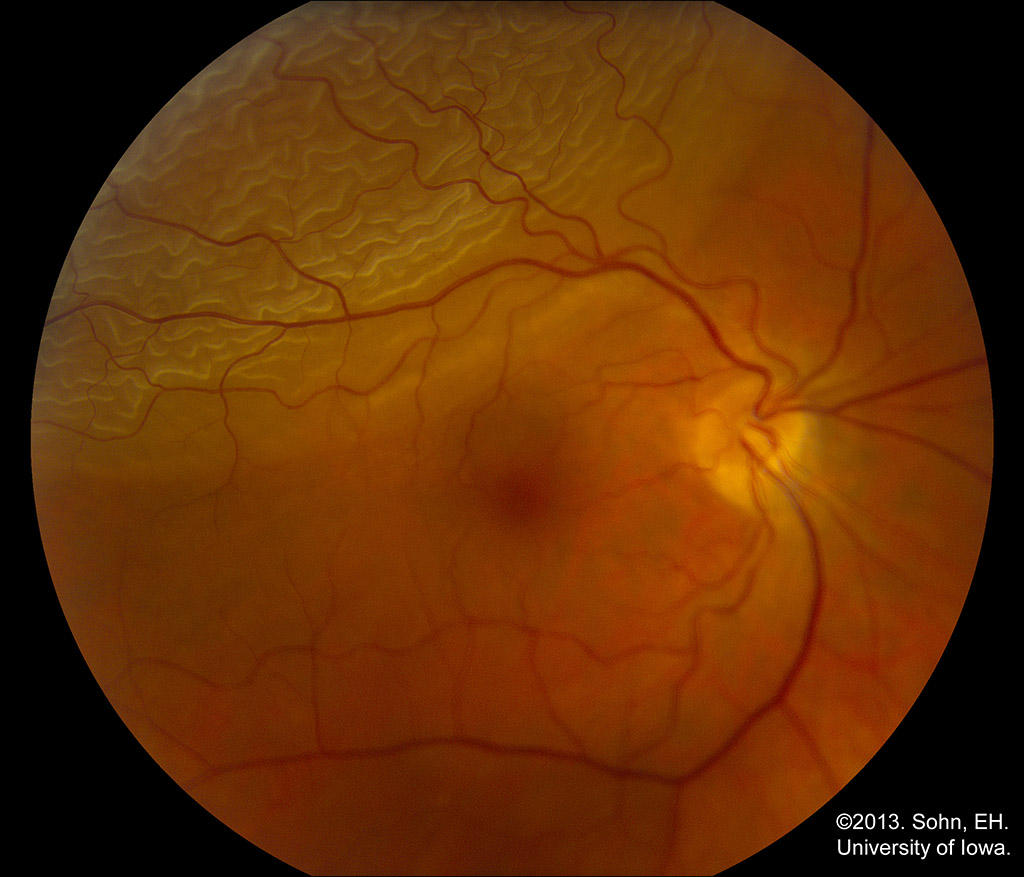
The primary way to diagnose a rhegmatogenous retinal detachment is by performing a proper fundus exam. When sitting the patient down at the slit lamp biomicroscope, prior to visualizing the fundus, you should look for Schaffer’s sign, a finding often present in patients with retinal tears and rhegmatogenous retinal detachments. Schaffer’s sign is positive when you use a thin beam of bright light without a lens and see fine pigment floating in the anterior vitreous. This pigment is circulating RPE cells released from a retinal break. When visualizing the fundus on the slit lamp or using indirect ophthalmoscopy, you will be checking for retina that appears separated from the normal structure of the eye. In the case of rhegmatogenous retinal detachment, you may see a large bullous separation of the retina. There will always be an associated tear or hole. These are often more peripheral and may be visualized better using an indirect ophthalmoscope.
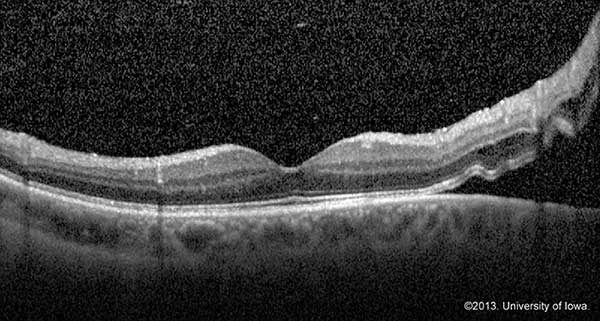
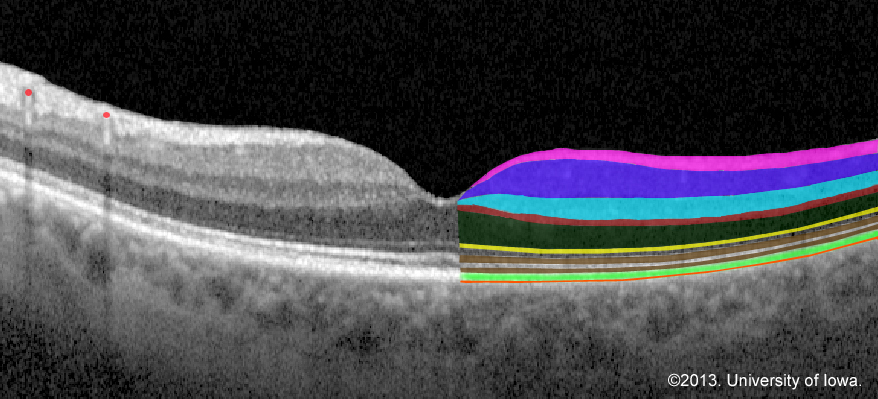
OCT allows for visualization of microscopic changes in the retina including small amounts of exudate underneath the retina. OCT is not the gold standard for identifying retinal detachments, but it can be extremely useful for identifying foveal status and tractional or exudative retinal detachments. If the status of the fovea is still in question despite a good history and exam, obtaining an OCT may support your diagnosis and determine time course for treatment as well as prognosis.
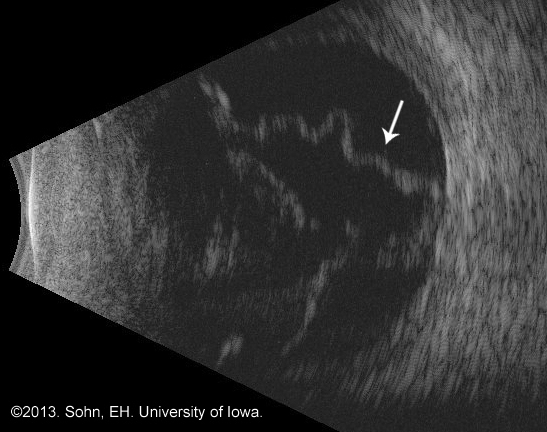
A B-scan is an ultrasound of the eye. If the media of the eye is not clear (e.g. from vitreous hemorrhage or severe cataract), ordering a B-scan is an excellent way to identify retinal detachments.
The treatment for retinal detachment depends upon the type and size of retinal detachment. For a small, peripheral RRD, the treatment of choice is cryotherapy of the break or laser photocoagulation around the detachment to prevent it from spreading. Once laser is applied, it takes between 3 and 14 days for maximal retinal adhesion to be achieved.(6) For a larger RRD, the primary treatment options are surgical. A scleral buckle, pars plana vitrectomy, or pneumatic retinopexy (all explained below) are the primary interventions used. Tractional retinal detachment is addressed most appropriately by treating the underlying disease, such as proliferative diabetic retinopathy. If traction threatens the macula, vitrectomy is usually indicated.5 Traction can also become severe enough to cause a tear or hole, becoming a RRD. Management of exudative retinal detachment involves treating the underlying disease (e.g. uveitis, choroidal tumor, etc.) to decrease the fluid between the neurosensory retina and RPE.
The status of the macula is the primary determinant of the emergency of the situation. If the macula, or more specifically the fovea, is still attached, the procedure is an emergency and should be performed within 24 hours. This is often referred to as a “mac-on” retinal detachment and is an emergency because central visual acuity is still preserved. Preventing the detachment from spreading to the fovea is extremely important. If the macula has detached it is referred to as a “mac-off” retinal detachment and prognosis for recovery of central acuity is worse, and thus urgent treatment is less critical. Nonetheless, studies show visual recovery is best if the procedure is performed within 7-10 days.(7-10)
The scleral buckle (SB) procedure was introduced in the 1950s(11) and involves the dissection of conjunctival tissue, exposure of the extraocular muscles, and wrapping a buckle underneath the extraocular muscles to support the sclera beneath the retinal detachment. Indirect ophthalmoscopy is used in coordination with this procedure to provide cryotherapy to the tears and to mark the sclera in the area of detachment. The marks are used so the buckle can be placed to provide support in the area of the detachment. The process by which this fixes the detachment isn’t completely understood. Changing the fluid dynamics underneath the detachment to allow the RPE to remove fluid, bringing the RPE closer to retina, and reducing traction are the primary ways this procedure is thought to work. The scleral buckle is often considered for RRD in phakic eyes. This is due to the fact that there is no significant difference in the proportion of primary reattachment between phakic eyes treated with SB and those treated with pars plana vitrectomy (PPV). In addition, scleral buckle treated eyes have greater 6 month postoperative best corrected visual acuity (BCVA) compared to eyes treated with vitrectomy. This is likely related to the increased incidence of posterior subcapsular cataracts in PPV treated phakic eyes.(12) However, regardless of lens status, the first time success rate for retinal reattachment with SB is above 80%.(13)
The pars plana vitrectomy was introduced in the 1970’s(14) and is considered by some to be superior to SB for the pseudophakic RRD. This procedure removes all vitreoretinal traction and allows for improved visualization of any associated retinal breaks/tears. In its most simple description, the vitreous is removed by introducing a needle through the pars plana that cuts and aspirates thousands of vitreous slices per second. As vitreous is removed, the eye is continually filled with balanced salt solution to maintain pressure. The final step involves introducing a medium into the eye (C3F8 gas, SF6 gas, or silicone oil) to hold the retina to the eye wall. Patient positioning based upon the location of the break and the medium used to fill the eye is required after the procedure. The first time success rate for retinal reattachment, regardless of lens status, is above 90%.(15)
Pneumatic retinopexy was introduced in 1986.(16) It is a minimally invasive procedure that can be accomplished in the clinic quickly. Therefore, this procedure can be a cost effective approach to retinal detachment. This procedure entails injecting a gas bubble (C3F8 or SF6) into the eye between the retina and the vitreous in an attempt to reapproximate the retina to the retinal pigment epithelium. However, the success rate for primary retinal detachment at two months for this procedure is approximately 66%.(17) Furthermore, depending upon the size and location of the break, there is a risk of separating the retina further if bubbles get underneath the detachment. Because a bubble is used, patient positioning is also required after the procedure. If failure does occur, it is usually early within the postoperative course, and re-operation (SB, vitrectomy) results in success rates also above 90%.(18)
The most common cause of post-operative failure in the affected eye is proliferative vitreoretinopathy. Other complications include induced myopia (SB), anterior ocular ischemia (SB), gas migration subretinally or to the anterior chamber (pneumatic retinopexy), cataract (vitrectomy and pneumatic retinopexy), and re-detachment (all types of treatment).
The fellow eye is also at an increased risk of retinal detachment. About 10% of phakic patients will have a retinal detachment in the fellow eye while 20-36% of aphakic patients will have a retinal detachment in the fellow eye.(3)
Suggested citation format
Strohbehn A. Sohn EH. Retinal Detachment: From One Medical Student to Another. EyeRounds.org. October 15, 2013; available from https://eyerounds.org/tutorials/retinal-detachment-med-students