Chief Complaint: Diplopia and "darkening of vision" in the right eye
Please pay particular attention to what is occurring to which eye: right (OD) or left (OS).
A 54-year-old gentleman noticed binocular vertical diplopia 4 months prior to presentation. The patient felt his symptoms were progressively worsening.
Furthermore, over the last week, while driving on the interstate, he noticed the vision in the RIGHT eye was starting to "darken."
During this four-month period, the patient was followed by an otolaryngologist for his sinus disease and nosebleeds. These symptoms had been present for the past 10 months. He was treated with cautery for the epistaxis, and ultimately, a polypectomy/anstrostomy was performed. On post-operative day #1, he notified the otolaryngologist that his diplopia was still present, although his nosebleeds and sinus disease had improved. One week post-operatively, the patient awoke with a "bulging" LEFT eye. This was accompanied by a LEFT facial droop. He was believed to have a Bell’s palsy and was started on doxycycline and prednisone 40mg daily by a local emergency room physician. The patient followed up with his otolaryngologist the next day, and the prednisone was increased to 80mg daily.
With no improvement after one week of the above treatment, he underwent decompression of the LEFT facial nerve. During these events, his diplopia persisted and he was referred to a neuro-ophthalmologist. The "darkening of vision" in the RIGHT eye remained throughout this entire time period.
The following is a summary of the laterality in this patient’s symptoms prior to presenting to us.
Past Ocular History: Presbyopia
Allergies: No known drug allergies
Family History: Non-contributory
Pupils: 4mm → 2mm, brisk, equal, 1.2 log units RAPD OD
Intraocular pressure: Applanation: 15 OD, 20 OS
Motility: Full, orthotropic OU
Confrontation visual field: Inferotemporal deficit OD, full OS
External: (Figure 1)
|
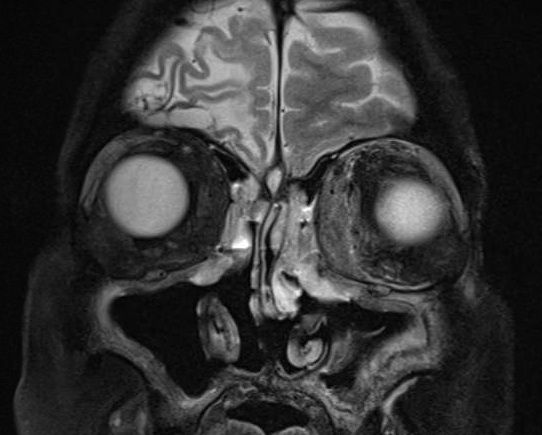 |
The patient underwent extensive examination and testing by multiple services including neuro-ophthalmology, oculoplastics, and otolaryngology. He initially underwent an endoscopic biopsy, which demonstrated acute and chronic inflammation with fibrosis.
In the meantime, the patient's visual acuity worsened to hand motion OD and 20/150 OS with worsening visual fields (Figure 6).
Given the pathology report above, the leading working diagnosis was idiopathic orbital inflammatory syndrome (IOIS); therefore, high dose steroids were started. The patient received methylprednisolone 1000mg intravenously for 3 days then started a slow oral prednisone taper starting at 80mg.
With steroid therapy, the patient's visual acuity and visual field testing initially improved over the short term.
However, over the next month, the patient failed to improve. If the diagnosis had been IOIS, we would have expected a rapid resolution of his symptoms.
At this point, the various involved medical teams re-evaluated the situation, which should always be done when the patient does not respond as expected to the management. The situation was discussed between the oculoplastic surgeons, neuro-ophthalmologists, and otolaryngologists, and the decision was made to re-biopsy the lesion.
An anterior orbitotomy was performed through a transcaruncular approach by the oculoplastics team.
The biopsy showed sclerotic tissue with a scattered chronic granulomatous inflammation with patchy infiltrates and necrosis. The differential diagnosis included ANCA-associated granulomatous vasculitis (Wegener’s granulomatosis) and idiopathic orbital inflammatory syndrome.
Lymphoma was ruled out with immunohistochemistry stains with CD20 and CD79a.
Of note, the more typical pathology seen in ANCA-associated granulomatous vasculitis (Wegener’s granulomatosis) shows occlusions of the vessels representing a vasculitis as well as interstitial granulomas. There are also polymorphic inflammatory infiltrates. The absence of these entities does not necessarily rule out the diagnosis of Wegener's granulomatosis.
Disease limited to the orbit alone or orbit and sinuses, such as in our patient, has been reported to show a more polymorphous pattern of infiltrative cells, which includes plasma cells, histiocytes, epithelioid cells and eosinophils compared to patients with systemic ANCA-associated granulomatous vasculitis (Ahmed 2008, Fecher 2002).
Laboratory work up was performed including an anti-neutrophil cytoplasmic antibody (cANCA) titer, which is 93% sensitive and 97% specific for systemic ANCA-associated granulomatous vasculitis (Wegener’s granulomatosis) (Nolle 1989). In limited orbital or orbital/sinus disease, the sensitivity is variable, between 32-67% (Nolle 1989, Woo 2001).
Our patient’s cANCA titer was > 1:80 (normal < 1:20)
He was referred to the rheumatology service and started on cyclophosphamide with the intention to transition to methotrexate, once in remission.
ANCA-associated granulomatous vasculitis* (Wegener's granulomatosis) is a granulomatous necrotizing vasculitis (Godman 1954). Godman and Churg described in a review the pathological features of this disease in the small arteries of the lungs and kidneys. In 1983, McClusky and Fienberg described independent areas of extravascular necrotizing granulomatous lesions that are now recognized as a classic pathologic finding (see above), especially in pulmonary tissue (McClusky 1983). The condition's eponym is named after Dr. Friedrich Wegener, who first described the disease in detail in 1936, although Peter McBride (1854-1946) in 1897 and Heinz Karl Ernst Klinger (born 1907) in 1931 reported similar disorders earlier.
Up to 50% of systemic ANCA-associated granulomatous vasculitis (Wegener’s granulomatosis) can have ocular/orbital involvement. Sixteen percent of patients will present with ocular inflammation. However, orbital or orbital/sinus involvement alone is rare. According to the largest study thus far, only 13 patients over 23 years (Ahmed 2008) have had disease limited to this area.
Of note, patients initially present with signs and symptoms, which can be extremely variable and nonspecific. Consequently, the diagnosis can be severely delayed.
In our patient, the diagnosis of ANCA-associated granulomatous vasculitis was somewhat delayed because of visits to multiple physicians in different areas of medicine. However, this is a not uncommon way of presentation. Because the initial presenting symptom may be highly nonspecific, a high index of suspicion is required. It also usually requires the professional collaboration of multiple medical teams including otolaryngologists, rheumatologists, pathologists, neuro-ophthalmologists, oculoplastic surgeons, as well as the patient's primary care physician.
The epistaxis our patient experienced initially was treated; however, the source of the presenting sign was not definitively determined. Furthermore, recall that his presenting symptom of decreased vision was in the RIGHT eye. Therefore, when he developed new symptoms in the LEFT eye, the working diagnosis needed to be readdressed and questioned.
In addition, when our patient did not respond as expected to steroid therapy alone, we questioned our leading working diagnosis at that time, which was IOIS. It is critical to reassess the situation if treatment does not deliver the expectant result.
Consequently, as mentioned above, many different medical departments worked together to re-evaluate the case. We collaborated with the otolaryngologists to perform the biopsy, first, in the least invasive manner with an endoscope. Because the pathology was inconclusive, a more invasive biopsy, then, was performed via an anterior orbitotomy. The ocular pathologist played a key role in diagnosing the disease as did the oculoplastics service, who performed the biopsyand ordered the laboratory testing. After the repeat biopsy and the blood work confirmed the diagnosis of Wegener's granulomatosis, our rheumatologists played an integral role in initiating the appropriate therapy promptly. This case is a great example of how multidisciplinary collaboration can lead to accurate diagnosis and prompt treatment.
EPIDEMIOLOGY:
|
SIGNS:Ocular:
Systemic:
|
SYMPTOMS:Ocular:
Systemic:
|
TREATMENTCyclophosphamide (first line of treatment for most in conjunction with corticosteroids.)
Once in remission switch to methotrexate or azathioprine. Corticosteroids can be tapered to a low maintenance dose, once remission is achieved. Plasmapheresis (in very severe disease, including pulmonary involvement or other end-organ damage) Alternative options in severe disease unresponsive to the above treatments:
|
Ahmed M, et al. Diagnosis of limited ophthalmic Wegener granulomatosis: distinctive pathologic features with ANCA test confirmation. Int Ophthalmol 2008;28:35-46
Bosch X, et al. Treatment of antineutrophil cytoplasmic antibody associated vasculitis: a systematic review. JAMA 2007; 298: 655-69
Carrington CB, et al. Limited forms of angiitis and granulomatosis of the Wegener’s type. Am J Med 1966;41:497-527
Fauci AS, et al. Wegener’s granulomatosis: prospective clinical and therapeutic experience with 85 patients for 21 years. Ann Intern Med 1983;98:76-85
Fecher FP, et al. Wegener’s Granulomatosis of the orbit: A clinicopathological Study of 15 patients. Laryngoscope 2002;112:1945-50
Godman GC, et al. Wegener’s granulomatosis: pathology and review of the literature. AMA Arch Pathol 1954;58:533-53
Leavitt RY, et al. The American College of Rheumatology 1990 criteria for the classification of Wegener's granulomatosis. Arthritis Rheum1990;33: 1101-7
McClsukey RT, et al. Vasculitis in primary vasculitis, granulomatoses and connective tissue diseases. Hum Pathol 1983;14:305-15
Nolle B, et al. Anticytoplasmic autoantibodies: their immunodiagnostic value in Wegener Granulomatosis. Ann Intern Med 1989;111:28-40
Woo TL, et al. Australasian orbital and adnexal Wegener’s study group. Australasian orbital and adnexal Wegener’s granulomatosis. Ophthalmology 2001;108:1535-1543
Woywodt A, Matteson EL. Heberden Historical Series: Wegener's granulomatosis--probing the untold past of the male behind the eponym. Rheumatology 2006;45:1303-6
* ANCA-associated granulomatous vasculitis has been proposed as a more appropriate and descriptive term for this condition rather than eponym of Wegener's granulomatosis. (Woywodt 2006)
Hong ES, Longmuir R, Syed NA, Allen RC. ANCA-associated granulomatous vasculitis(Wegener's Granulomatosis). Eyerounds.org. December 7, 2010; Available from: http://www.EyeRounds.org/cases/126-ANCA-associated-granulomatous-vasculitis.htm

Ophthalmic Atlas Images by EyeRounds.org, The University of Iowa are licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported License.